Rare Disease Day is on February 29th and is a day dedicated to raising awareness for patients, families and carers around the world that are affected by rare diseases. Sean Wachter is an incredible rare melanoma survivor from the United States who has used his story and his passion for wrestling to give back and inspire others. We thank Sean for sharing his story with us.
Can you describe your journey with stage 4 rare melanoma?
Sean Wachter:
The actual receiving my diagnosis was not difficult, it was getting to the point of someone finally giving me a diagnosis that was the difficult part. I spent a period from about March of 2016 to right up until when I got sick on September 26, 2016 visiting various doctors and telling them that something wasn’t working with my body correctly. I had unfortunately been in a really bad accident about three or four years prior where I broke my neck, back and lost my ability to walk, and once I had regained my ability to walk unassisted was when things started feeling off in my body. Every doctor I went to said “It’s from your accident. This is post concussion syndrome” – they wanted to write it off.
Around May of 2016, I went on a date and while we were sitting there I got very dizzy and collapsed while eating. I was taken by ambulance to a local medical facility and they said I was just dehydrated and sent me on my way. About a month later, I was at a Mets baseball game and I blacked out right in the middle of the concourse. I wound up going to two separate hospitals that evening, because I wasn’t satisfied with the level of care I was getting at the first one. But the second one said the same thing, that I was dehydrated and had the flu so I was sent home again. About a month later, the same thing happened and I was taken to a hospital in New York City. So now I was starting to deal with good, world renowned hospitals but I still wasn’t taken seriously.
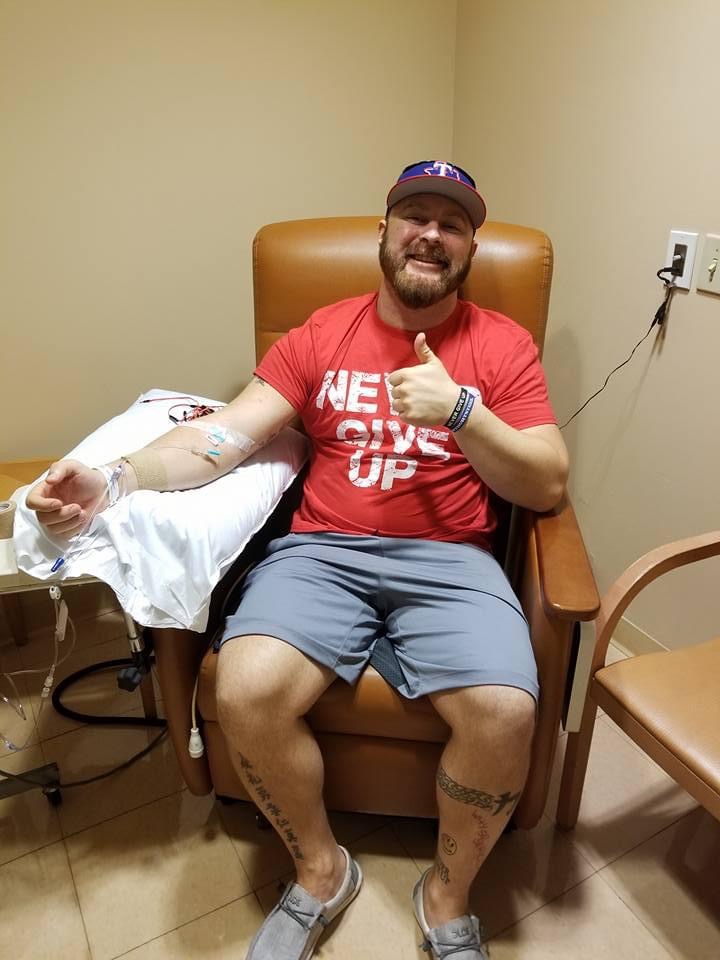
In between these bigger incidents I had also gone to my local medical facility about six times telling them “Hey, something’s not right. Please listen to me”. The next time I went back was September 23, 2016 and I was not treated very well. I was basically told “You’re either drunk or hungover” but that was impossible because I didn’t drink. I didn’t know it at the time, but they were actually going to put me on a psychiatric hold – they thought I was making it all up in my head so they were going to hold me overnight for observation. I started to argue with them then, and pushed to see if they could give me an MRI but they refused because they thought nothing was wrong. Essentially, all they were willing to do was give me a bag of fluid so I ended up leaving on my own and going to my parents house because I knew something was wrong but I didn’t know what yet.
My parents had to leave for a few hours that weekend and within 10 minutes of them being gone, I was on the floor throwing up, my face was drooping and my legs wouldn’t work. Luckily, I managed to call my Dad and he came back and sprung into action. He took me back to the hospital but they didn’t want to help me again – they just said “Oh he’s back”. I proceeded to throw up all over myself, and they seemed more aggravated than anything because they were asking me to fill out the insurance forms but I couldn’t use my hands properly. I got put into the non-emergency area of the hospital and I asked for a CT scan but the nurse practitioner said I was too young. A few minutes after that, I started having a stroke, which unfortunately my parents had to see. That was when we found out I had a golf ball sized tumor in my cerebellum that caused a massive, massive hemorrhage. A doctor took my Dad aside while I was in surgery and told him I had late stage melanoma. I regained consciousness two days after the surgery, and I remember my dad just rubbing my ankle and telling me the news. And I said to him, “Okay, we’re gonna fight this thing and we’re gonna beat it”.
I was in the hospital for about a month and the people around me really showed up. My high school and college football teammates wrote up a spreadsheet of who was going to see me and what they would bring. I went to Sloan Kettering Hospital and I had radiation to clean up the surgical bed. When I was getting cleaned up, I noticed something that really influenced my cancer journey. At the time, they didn’t have a separate pediatric unit for radiation and I looked around and I saw a lot of sick kids that were maybe two or three years old. And I just thought “I’m not gonna sit here and feel sorry for myself. I’ve made every mistake a 31 year old man can make, and these kids haven’t even had a chance to steal a cookie yet”. So at that moment, I decided that whatever happens, happens. I came across an American former football player whose name is Inky Johnson, and he was speaking about how it’s not always about you, it’s about the people who come to lift you up. And I think that sometimes as a cancer patient or cancer survivor, it gets lost on us that while it may seem like we’re in the middle of the ocean surrounded by sharks by ourselves, in actuality there are still people around us, and we have a responsibility to them too. So the “It’s not about you” really resonated with me.
I continued to go on with my life, and we tried different immunotherapies and chemotherapies that unfortunately caused my liver to keep crashing. I went to the ICU and was told that I had a rare complication called lepto meningitis disease where the cancer spreads to your spinal cord fluid, and essentially the cells start to coagulate and you lose your brain function. I was told I had 12 weeks to live and to get my affairs in order. That was a right hook that sent me to my knees real quick. But I’m stubborn and didn’t want to accept it.


With great research and clinical trials, we were able to find out that I had certain mutations that I could use to my advantage and that pretty much stopped the progression. All of a sudden, I started getting more and more time, which was really amazing. Around April of 2017, my doctor asked if I wanted to try another infusion on top of the two pills I was taking already. And that actually started to send the future scans into regression – I was having one of the best responses you could have to the medication. I decided I wanted to do something more, and I began coaching high school football. It was cool because me being sick really held the kids accountable. If a guy showed up late to practice, you’d have someone on the team saying “He was in chemo today, but you’re late?”. So “What’s your excuse? ” became their rallying cry. It was amazing because at one of the games, my friend arranged for all of the kids to wear these shirts that said Long Beach Football on the front and Coach Wachter Strong with the melanoma ribbon and the saying “What’s your excuse” on the back. It makes me cry to this day.
My scans were still regressing at that point and no one could really make sense of it. At that point, my parents talked to me and suggested I get back out there, because I’d fallen apart physically – I used to be in great shape but I lost that. So I put myself out there and started going on dates, and after a few attempts I saw my current wife on Bumble. I recognized her through a mutual friend and we started talking on Facebook. I asked her on a date and we spent 6 hours just talking. When I told my mom about her she actually said that we were born 5 houses apart, so she was the first girl I ever hung out with! Within a few months, we moved in together and I started helping her raise her daughter, which has been the biggest blessing. I have done so many cool things, but raising my stepdaughter has been the coolest thing I’ve gotten to do. She’s my best friend. I have no problem telling people my best friend’s eight year old because it’s her and my wife who made me realize I was not only going to beat cancer, I was going to go through it in a spectacular fashion.
I decided to go into business and open an independent coffee chain on Long Island, both to show myself I could and to leave my wife and stepdaughter with something just in case. I always tell people that the coolest thing about doing the coffee shop was that it showed me what I still had in the tank because I would go from six o’clock in the morning to one o’clock in the morning working at our three locations. It’s cool to say I got to run a company from a chemo chair. Sometimes people treat cancer patients like they are totally disabled, so that showed me I was still able and that no matter the circumstances I needed to keep living my life.
In the summer of 2021, I got some more scans done and they showed further regression without treatment. So I took my last IV infusion of immunotherapy and got to ring the bell, which was awesome. I was still taking the oral chemo pills, so I wasn’t fully good yet but my scans and spinal taps kept coming out positively so in 2022 the doctors asked if I wanted to stop taking the chemotherapy pills.
It was very fitting for me because in March of 2022 my wife, my stepdaughter, and I were going to see a WWE live event in Connecticut. I absolutely love wrestling so I rented a limousine for us and got premium tickets. When we were on the way, my phone rang, and I saw the number of the hospital. I didn’t know if I wanted to answer it right then and there just in case but I begrudgingly picked it up, and I got a “Congratulations. As of right now you are the only documented survival case of this type of cancer”. I was crying happy tears and I got to tell my wife and stepdaughter. It was amazing.
My buddies wanted me to throw a party, but I did feel some survivor’s guilt so I decided to throw a fundraiser for Memorial Sloan Kettering Kids. It was a wrestling fundraiser and one of my friends had this brilliant idea of putting a poll on Facebook, essentially saying “If Sean wrestles, will you donate more money?”. I wrestled and it was a big success. Since then, I married my wife and we had our miracle baby. We also hosted another fundraiser where I wrestled in honor of my friend who passed away. It was such a wonderful event, it was twice as big. It was attended by local politicians and the press did an article on me that I put it on my LinkedIn, and from that ESPN found me. From there, I started wrestling a lot more and now we’re working on some amazing stuff. It’s been a crazy ride and I think my story really is a testament to folks that there’s life during and after cancer. I don’t think anyone should let their cancer diagnosis get in the way of them continuing to make their plans and live their lives. Of course that’s easier said than done but something as simple as getting out of the bed in the morning is still a victory over cancer. To me, without cancer there’s no wrestling and I realize that I have this responsibility to keep spreading awareness and sharing my story. I just hope that if somebody hears my story or sees me wrestle, they’re able to live a little bit fuller, or fight a little bit harder. I’d go down the same road 1000 times again, if it helps one person out.















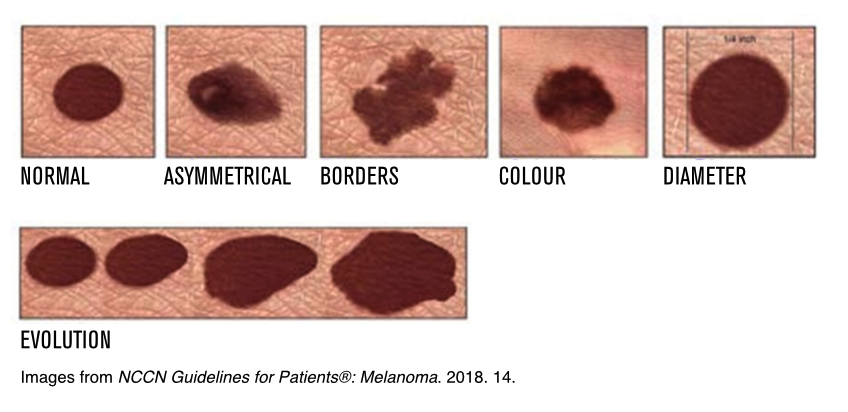 A common and effective tool to help you spot melanoma is the ABCDEs of Melanoma:
A common and effective tool to help you spot melanoma is the ABCDEs of Melanoma: